Introduction
Coughing is an important respiratory defense reflex that clears foreign objects and invading pathogens from the airways. At the same time, coughing is a common symptom in many respiratory diseases, such as asthma and post-COVID syndrome. Although coughing is a protective reflex for the airways, it becomes excessively activated in pathological states, leading to chronic coughing. Current treatment strategies are unable to fully resolve chronic cough, which significantly impacts patients’ quality of life. Most cough research has been conducted using guinea pigs, cats, and other animals, which are costly to breed and lack effective genetic tools. The use of mice as experimental animals provides more controllable genetic tools for studying cough. However, as of now, there is no clear cough behavior paradigm in mice. Additionally, the central neural circuitry controlling cough remains unexplored, and understanding this mechanism could provide new insights into the pathology of chronic cough.
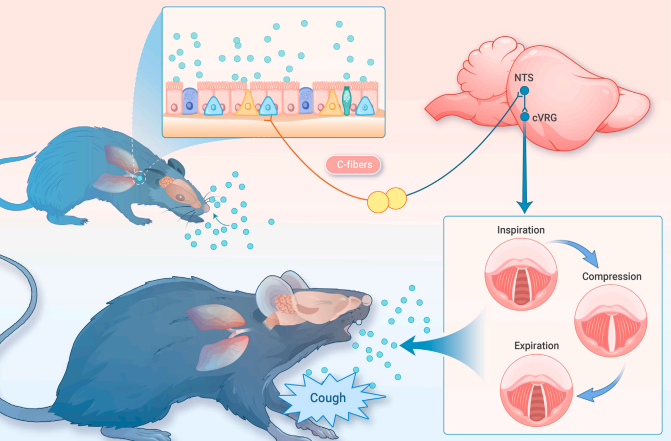
On November 4, 2024, Peng Cao’s laboratory at the Beijing Institute of Life Sciences / Tsinghua University Joint Biomedical Research Institute published a research paper titled “Brainstem opioid peptidergic neurons regulate cough reflexes in mice” in The Innovation. The study developed a novel three-channel cough behavior paradigm that simultaneously monitors intrapleural pressure (IPP), box airflow, and ultrasound, allowing differentiation between cough and sneeze—two distinct respiratory defense reflexes. Using this paradigm, the researchers identified a specific population of opioid peptidergic (Pdyn) neurons in the nucleus tractus solitarius (NTS) that project to the central respiratory rhythm generator (cVRG) and are critical for the cough reflex in mice.
Development of a Quantifiable Mouse Cough Behavior Paradigm
The researchers first developed a quantitative paradigm to analyze mouse cough behavior. By using whole-body plethysmography (WBP), an in vivo telemetry device, and an ultrasound microphone, they were able to monitor the airflow (Boxflow), intrapleural pressure (IPP), and subtle sound changes (sonogram) in real time. This system successfully induced two distinct airway defense reflexes, Class I and Class II, in response to capsaicin, with each reflex having different sound frequencies and durations. Since the nerve endings of the glossopharyngeal (NG) and trigeminal (TG) nerves primarily innervate the larynx and nasal mucosa, the researchers used chemogenetic inhibition viruses (hM4Di) to selectively or simultaneously inhibit the Trpv1+ neurons in the NG and TG. This inhibition reduced both Class I and Class II reflexes, with Class I being identified as the cough reflex.
Identifying the Role of Pdyn+ Neurons in the Cough Reflex
By using capsaicin to stimulate FosTRAP2 mice, the researchers pinpointed the dorsal vagal complex (DVC) as the brain region responsible for physiological cough induced by capsaicin. Through fluorescence in situ hybridization (FISH) staining of the NTS, they discovered that three opioid peptides—prodynorphin (Pdyn), proenkephalin (Penk), and proopiomelanocortin (Pomc)—were specifically expressed in the DVC. Chemogenetic inhibition of Pdyn, Penk, and Pomc neurons in the DVC revealed that Pdyn+ neurons are crucial for mediating the cough reflex. In particular, inhibiting Pdyn+ neurons significantly reduced cough triggered by capsaicin or citric acid.
Calcium Imaging of Pdyn+ NTS Neurons During Cough
Using AAV-DIO-GCaMP and fiber-optic implantation in Pdyn-Cre mice, the researchers used fiber-optic recording to observe calcium signals from Pdyn+ neurons in the NTS. They found that the respiration pattern was coupled with GCaMP signals, and coughing caused a significant rise in GCaMP signals in Pdyn+ NTS neurons, while sneezing had no noticeable effect. To further confirm that the GCaMP signal rise in Pdyn+ NTS neurons was induced by peripheral Trpv1+ vagal nerve input, they performed unilateral vagotomy or used Trpv1 knockout (KO) mice for fiber-optic recording. Both vagotomy and Trpv1 KO diminished the GCaMP signal rise induced by coughing, confirming that Pdyn+ NTS neurons receive and respond to signals from Trpv1+ vagal afferents.
Activation of Pdyn+ NTS Neurons Induces Cough Reflex
To determine whether activation of Pdyn+ NTS neurons is sufficient to trigger coughing, the researchers used chemogenetics to activate Pdyn+ NTS neurons by injecting AAV-DIO-hM3Dq into the NTS of Pdyn-Cre mice. Activation of these neurons induced coughing without altering the mouse’s respiratory rate, and the induced cough exhibited similar parameters to those triggered by capsaicin. Additionally, the researchers used optogenetics, injecting AAV-DIO-ChR2 into the NTS of Pdyn-Cre mice and observing that optogenetic activation of Pdyn+ NTS neurons also effectively triggered coughing. These results confirm that activation of Pdyn+ NTS neurons is a sufficient condition to induce the cough reflex.
Identifying the Downstream Pathways of Pdyn+ NTS Neurons
To investigate the downstream pathways of Pdyn+ NTS neurons, the researchers injected AAV-DIO-EGFP-Syb2 into the NTS of Pdyn-Cre mice. They found that EGFP+ terminals were primarily located in the cVRG and the ipsilateral intermediate reticular nucleus (IRt). To examine the roles of the Pdyn+ NTS-cVRG and Pdyn+ NTS-IRt pathways in the cough reflex, the researchers used chemogenetic inhibition (hM4Di) and optogenetic activation (ChR2) in these regions. Inhibition of the Pdyn+ NTS-cVRG pathway significantly reduced capsaicin-induced coughing, suggesting that this pathway is necessary for the cough reflex. Additionally, optogenetic activation of the Pdyn+ NTS-cVRG terminals reliably induced coughing.
Synaptic Connections Between Pdyn+ NTS-cVRG Pathway and Cough-Related Muscles
Finally, to explore whether the Pdyn+ NTS-cVRG pathway synaptically connects to cough-related muscles, the researchers used AAV-DIO-EGFP-Syb2 to label Pdyn+ NTS axon terminals and pseudorabies virus (PRV) to retrogradely label neurons in the diaphragm, intercostal muscles, and abdominal muscles. They observed that EGFP+ Pdyn+ NTS axon terminals in the cVRG were scattered among the dsRed+ PRV-labeled neurons, indicating a synaptic connection. These morphological results suggest that the Pdyn+ NTS-cVRG pathway may regulate the cough-related muscles via synaptic interactions, thereby controlling the cough reflex.
Conclusion
In summary, this study developed a novel three-channel monitoring paradigm for mouse cough behavior and, using this paradigm, identified a specific neural circuit regulating the cough reflex: the Pdyn+ NTS-cVRG pathway. This finding lays a crucial foundation for further research into the deep neural mechanisms underlying coughing.
Corresponding Author: Senior Researcher Peng Cao, Beijing Institute of Life Sciences / Tsinghua University Joint Biomedical Research Institute.
Co-first Authors: Hai-Cheng Lu (2022 TIMBR PhD student), Wen-Xuan Zheng (2022 PTN PhD student), Postdoctoral Researchers Guo-Qing Chen, Miao Zhao, Hua-Ting Gu, Mei-Zhu Huang, Master’s Student Xia-Ting Li (Capital Medical University).
This research was funded by the Beijing Municipal Science and Technology Commission, the Tsinghua University Joint Biomedical Research Institute, and the Beijing Institute of Life Sciences.
Paper Link: https://www.cell.com/the-innovation/fulltext/S2666-6758(24)00159-0

