Parkinson’s disease, as a highly prevalent neurodegenerative disease worldwide, is characterized by the abnormal aggregation of α-synuclein (α-Syn), which forms Lewy bodies and Lewy neurites. These aggregates progressively spread, causing neuronal damage, and become a key factor in the disease’s progression. However, for a long time, the scientific community lacked the technical tools to accurately and real-time track α-Syn aggregates in vivo, hindering clear observation of their dynamic propagation and in-depth analysis of their pathological effects. This limitation severely restricted research on the pathogenesis of Parkinson’s disease and the development of related therapeutic approaches.
On March 4, 2026, a significant research breakthrough was published online in the prestigious journal Cell. Led by Professor Peng Cao’s team from the Beijing Institute of Life Sciences/Tsinghua University Institute for Biomedical and Cross-Disciplinary Research, in collaboration with multiple research institutions including the Institute of Biophysics, Chinese Academy of Sciences, University of Science and Technology of China, Peking University, and Beijing Normal University, the team successfully developed a novel genetic-encoded fluorescence probe system and established a corresponding gene knockout mouse model. This achievement marked the first real-time, highly specific, and sensitive visualization of α-Syn aggregates in the living brain, and it systematically elucidated the pathological effects of these aggregates in different neuronal subtypes. This provides a revolutionary tool for the study of Parkinson’s disease and other synucleinopathies, opening up new paths for mechanistic exploration and therapeutic development.

 To address the challenge of in vivo detection of α-Syn aggregates, the research team innovatively designed a fusion protein strategy. They fused mouse-derived α-Syn with two fluorescent proteins, EGFP and tdTomato, through linker peptides for co-expression. After screening and functionally validating seven variants of linker peptides, the team identified the optimal 6H linker peptide. Using this, they constructed the α-Syn-6H-EGFP and α-Syn-6H-tdT reporter genes. The probe system demonstrated a unique aggregation-dependent fluorescence enhancement feature: when α-Syn existed in its monomeric form, the fluorescence signal was weak, but once α-Syn aggregates were formed, the fluorescence intensity increased by fivefold. Moreover, the system showed excellent biocompatibility, not inducing abnormal aggregation of α-Syn nor interfering with the normal physiological functions of endogenous α-Syn. The co-localization ratio with phosphorylated α-Syn reached 93%-97%, achieving an internationally leading level of detection precision. Using CRISPR/Cas9 gene editing technology, the team further developed Snca-6H-EGFP and Snca-6H-tdT gene knockout mice. In these models, the expression patterns of the reporter genes matched those of endogenous α-Syn, providing an ideal animal model platform for the study of α-Syn aggregates in vivo.
To address the challenge of in vivo detection of α-Syn aggregates, the research team innovatively designed a fusion protein strategy. They fused mouse-derived α-Syn with two fluorescent proteins, EGFP and tdTomato, through linker peptides for co-expression. After screening and functionally validating seven variants of linker peptides, the team identified the optimal 6H linker peptide. Using this, they constructed the α-Syn-6H-EGFP and α-Syn-6H-tdT reporter genes. The probe system demonstrated a unique aggregation-dependent fluorescence enhancement feature: when α-Syn existed in its monomeric form, the fluorescence signal was weak, but once α-Syn aggregates were formed, the fluorescence intensity increased by fivefold. Moreover, the system showed excellent biocompatibility, not inducing abnormal aggregation of α-Syn nor interfering with the normal physiological functions of endogenous α-Syn. The co-localization ratio with phosphorylated α-Syn reached 93%-97%, achieving an internationally leading level of detection precision. Using CRISPR/Cas9 gene editing technology, the team further developed Snca-6H-EGFP and Snca-6H-tdT gene knockout mice. In these models, the expression patterns of the reporter genes matched those of endogenous α-Syn, providing an ideal animal model platform for the study of α-Syn aggregates in vivo.
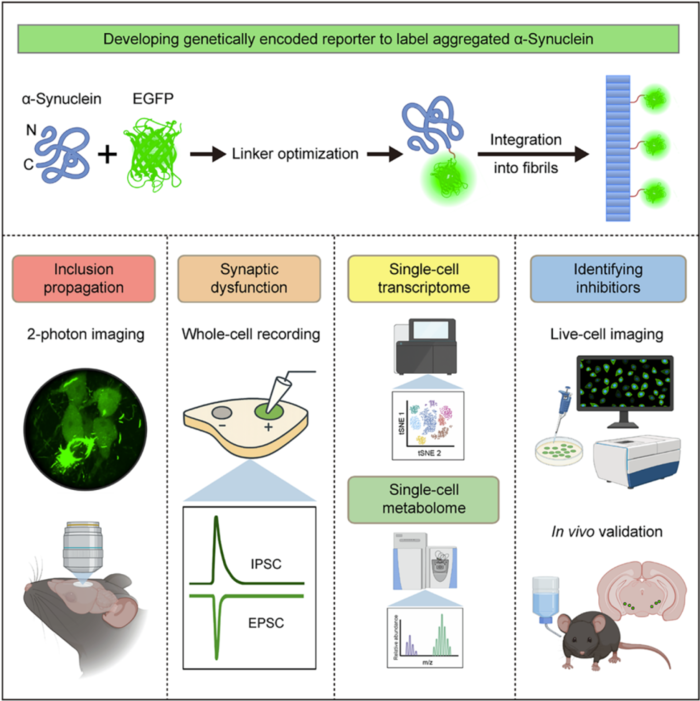
The α-Syn aggregate fluorescent probe developed in this study can be widely applied in live-cell imaging and recording, single-cell omics analysis, and functional drug screening.
The developed α-Syn aggregate fluorescence probe has broad applications, including live-cell imaging, single-cell transcriptomics analysis, and functional drug screening. With this innovative tool, the research team made several groundbreaking scientific discoveries. In the study of the mechanism of α-Syn aggregate propagation, the team injected pre-formed α-Syn fibrils into the prefrontal cortex and associative cortex of knockout mice and, using two-photon in vivo imaging, captured the diffusion process of α-Syn aggregates in the brain of awake mice for the first time. After 28 days post-injection, the aggregates were still confined to the injection site, but by day 84, the aggregates had spread to multiple brain regions, including the primary motor cortex, secondary motor cortex, and primary somatosensory cortex. This discovery provides direct in vivo experimental evidence for the prion-like propagation hypothesis of α-Syn aggregates, filling a critical gap in the field.
In terms of pathological functional analysis, the team performed in-depth studies combining Ca²⁺ imaging and whole-cell patch-clamp techniques. They found that in primary motor cortex neurons carrying α-Syn aggregates, the frequency of Ca²⁺ transients in the resting state was significantly reduced, with 21% of neurons losing activity, and the firing frequency of spontaneous action potentials also significantly decreased. In dopaminergic neurons in the substantia nigra, α-Syn aggregates led to a reduction in the amplitude of both excitatory and inhibitory postsynaptic currents. Further single-cell transcriptomic sequencing and mass spectrometry analysis confirmed that α-Syn aggregates severely disrupted mitochondrial function, the ubiquitin-proteasome system, lysosomal function, and other cellular pathways, ultimately causing dopamine metabolism disorders. This result clearly elucidates the molecular mechanisms by which α-Syn aggregates damage neuronal function.
To achieve specific research on α-Syn aggregates in different neuronal subtypes, the team developed a Cre recombinase-dependent RCL-Snca-6H-EGFP mouse model. By crossing this model with various neuronal subtype-specific tool mice, the team successfully achieved specific labeling of α-Syn aggregates in dopaminergic, glutamatergic, and cholinergic neurons. Regarding the common non-motor symptoms of Parkinson’s disease, the team found that when α-Syn aggregates appeared in cholinergic neurons of the dorsal motor nucleus of the vagus nerve, the excitability of these neurons was significantly reduced, leading to gastrointestinal motility disorders. This discovery provides direct experimental evidence for the pathogenesis of non-motor symptoms in Parkinson’s disease and lays the foundation for the research and intervention of these symptoms.
The reporter gene system developed in this study also has efficient drug screening capabilities, making it an important platform for Parkinson’s disease drug development. The team used this system not only to verify the inhibitory effects of known α-Syn aggregation inhibitors, such as heparin and squalamine, but also to discover, for the first time, that the potassium channel inhibitor 4-aminopyridine (4-AP) significantly inhibits α-Syn aggregation. The mechanism of action was found to be through the enhancement of neuronal action potential firing, which inhibits the abnormal aggregation of α-Syn. In vivo experiments showed that 4-AP treatment effectively reduced the content of α-Syn aggregates in the substantia nigra and dorsal motor nucleus of the vagus nerve, while significantly alleviating Parkinson’s disease-related motor dysfunction and gastrointestinal abnormalities. This discovery reveals a bidirectional regulatory relationship between neuronal electrical activity and α-Syn aggregation, providing a new therapeutic target for Parkinson’s disease and offering important candidate molecules for the development of new treatments.
This research exemplifies the success of interdisciplinary collaboration and has successfully overcome the technical bottleneck in the study of α-Syn pathology in the living brain. The probe system developed has high specificity, sensitivity, and excellent biocompatibility, and its applications extend beyond Parkinson’s disease to other synucleinopathies such as Lewy body dementia and multiple system atrophy. Currently, the related gene knockout mouse models and reporter gene vectors are available for global researchers through the Peng Cao Laboratory, providing strong technical support for neuroscience research worldwide. Furthermore, the α-Syn aggregation regulation pathways revealed by this study, as well as the promising therapeutic potential of 4-AP, offer new ideas for targeted therapy and core candidate drugs for Parkinson’s disease, with significant value for basic research and clinical translation.
The co-corresponding authors of this research paper include Professor Peng Cao from the Beijing Institute of Life Sciences, Professor Xiaoqun Wang from the Institute of Biophysics, Chinese Academy of Sciences, Professor Wei Xiong from the University of Science and Technology of China, Professor Zhuan Zhou from Peking University, and Professor Qian Wu from Beijing Normal University. Researchers Li Zhang, Minhui Yu, Guoqing Chen, Siyuan Ge, and Mengdi Wang are co-first authors of the paper. Professor Fengchao Wang’s team at the Transgenic Animal Center provided critical technical support for this work, while Professor Zhihui Liu’s team at the Institute of Biophysics made significant contributions.
It is important to emphasize that this research took nine years to complete, with more than ten graduate students and postdoctoral researchers from the Beijing Institute of Life Sciences participating in the project. This work was supported by the institute’s long-term dedication to fostering a focused and distraction-free research environment for scientists.
https://www.cell.com/cell/fulltext/S0092-8674(26)00163-7

